Curing Cancer Relies on Genome Map Guiding Medical Care
By John Lauerman
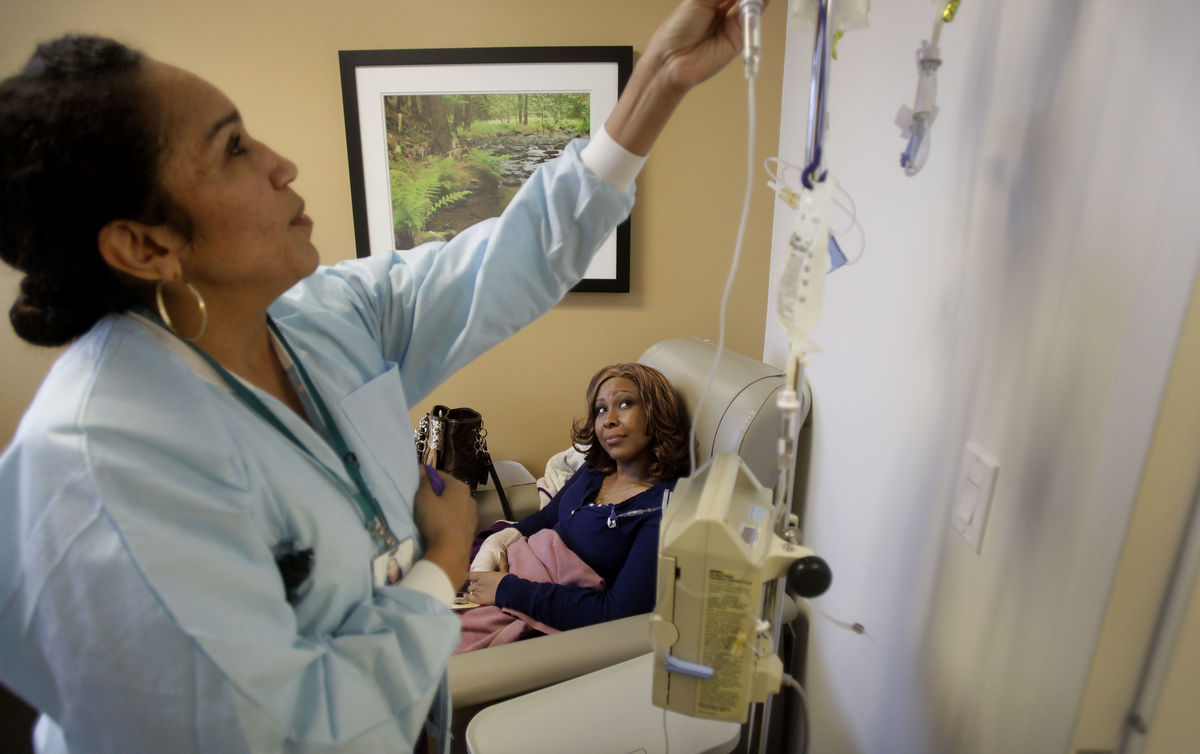
Kristal James, a medical technician in suburban Dallas, spent more than a year fighting rapidly spreading breast cancer that looked like it might take her life. As doctors raced to save her, they decided to sequence her tumor’s genome.
The sequencing showed surprising abnormalities in her diseased tissue’s DNA, confirming that a new drug they were trying was targeting genetic aberrations in her tumor. James, 33, has since been able to return to attending her son’s sporting events and school meetings.
“I can be a mommy again,” she said a year later. “It’s a different life than a year ago. For me, it’s making up for time I missed.”
A decade after the first draft of the human genome was published, hospitals and clinics are using DNA sequencing to generate better treatments and diagnoses for patients with rare childhood diseases, cancers and other mysterious conditions. Using new technology that can effectively print out an individual’s genome -- the instruction manual for making all the body’s cells -- doctors are examining individual components, called bases, to slow intractable cancers and treat one-of-a-kind diseases in children.
“This is going to be transformative to medicine,” said John Niederhuber, former director of the U.S. National Cancer Institute from 2005-2010, and now executive vice president of the Inova Health System hospital chain in northern Virginia.
Genome Sequencing
Doctors gave life-saving treatment to a 6-year-old after an exploration of his genes in 2009 indicated that a bone-marrow transplant might stop severe intestinal inflammation. Inova’s Virginia hospitals are sequencing the genomes of 500 premature babies and their parents to see what kind of information can be gleaned for improving care.
A California research center is sequencing the full genomes of 1,000 healthy people ranging in age from 80 to 108 years old to determine how to promote disease-free aging. In December, the U.S. National Human Genome Research Institute began funding projects designed to help doctors learn how to use genomic data to treat patients.
Using faster, more accurate technologies, doctors are combing through ever-wider swaths of the human genome to pinpoint and counteract the causes of disease. Cancer is among the most promising targets because it is essentially a disease in which damaged genes let cells grow without restraint, said Barrett Rollins, chief scientific officer of the Harvard University-affiliated Dana Farber Cancer Institute in Boston.
Deciphering Genes
Dana Farber now offers every patient who walks through the door a test for almost 500 gene variations; in about six months, that program will expand so that patients will have access to the deciphering of all their genes.
“If you’re working at a cancer center and not thinking about this, you should be fired,” Rollins said.
While the price of genome sequencing is falling to $1,000 and research money will sometimes pay for these procedures, they aren’t routinely covered by insurance. Some patients are paying out of pocket to have their genomes sequenced, and not all of them can be helped by the procedure because their tumors have so many genetic abnormalities.
“We have patients who walk in and say ‘I paid $50,000 to sequence the genome of my cancer,’” Niederhuber said at a November conference at Harvard Medical School.
Steve Jobs’s Genome
Steve Jobs, the late Apple Inc. chief executive officer, spent $100,000 to have his cancer genome sequenced in an effort to defeat a rare malignancy called neuroendocrine tumor. Jobs also underwent a liver transplant in 2009 to stop the spread of the disease. He died at the age of 56, eight years after being diagnosed with cancer.
While gene sequencers around the world churn out millions of gigabytes of genetic data, most of the human genome remains unexplored and incomprehensible even to the savviest scientists, let alone practitioners caring for families. With so much unknown, sequencing doesn’t hold answers for every patient.
Insurers are still waiting for more information about when and for whom sequencing the whole genome is an effective procedure, said Susan Pisano, spokeswoman for America’s Health Insurance Plans, a Washington-based industry group.
“Scientific evidence drives coverage,” Pisano said in a telephone interview. “You want to make sure that it’s an effective procedure, a safe procedure, and you want to know what you’re going to do with the results. And these are all questions that are still being asked.”
Shaping Treatment
For Kristal James, the mom in Dallas, the procedure helped shape her cancer treatment. In January, 2010, she took a few hours off from work to meet with the surgeon scheduled to perform her mastectomy at Baylor Plano Medical Center. She’d already had lumps removed from her breast twice and taken anticancer drugs. In the waiting room, she chatted with a woman who had undergone the procedure. The woman said the surgeon, Lynn Canavan, had a “light touch” that minimized pain.
James felt healthy and confident when her surgeon entered the office for their appointment. When James’s MRI image came up on the screen, both of them were seeing it for the first time. Canavan did a double-take; in just a few weeks, small colonies in James’s breast had grown as big as 10 centimeters, or about 4 inches.
“What do I do?” James asked the surgeon. “What’s going to happen?”
“You have to go to your oncologist and give him this,” Canavan replied.
Feeling Despair
Canavan told James she would probably need another round of powerful chemotherapy along with radiation treatment and the mastectomy. It was one of the few times she could recall feeling despair.
“I couldn’t talk,” James said. “I sat there in stunned silence and the tears were just running. It felt like we had moved backwards, and then even further back.”
Her prospects continued to spiral downward. Doctors discovered tumors in her left breast. She developed swelling in her side. James’s doctors referred her to Joyce O’Shaughnessy an oncologist at Baylor University Medical Center in Dallas.
O’Shaughnessy specializes in “triple negative” breast cancer that doesn’t respond to surgery, drugs and radiation. She was participating in a national study to see if gene sequencing could help identify new therapies for the hardest-to-treat breast cancers.
Tumor DNA
James’s tumor DNA was sent to the Translational Genomics Research Institute in Phoenix for sequencing on equipment made by Life Technologies Corp. Meanwhile, O’Shaughnessy put James on an experimental regimen of Novartis AG’s Afinitor, a drug normally used to stop the immune system from rejecting transplanted organs, as well as treating brain and kidney cancer. James also took Roche Holding AG’s Avastin, which shuts down blood-vessel formation in tumors, and taxol, a hormone blocker often used against breast cancer.
Novartis fell 0.9 percent to 51.60 Swiss francs at the close in Zurich trading. Roche dropped 0.1 percent to 163 francs.
James said she could feel her tumors shrinking. Within a few weeks she felt that hard cancer tissue was disappearing from beneath her skin.
In the meantime, the sequencing showed that James’s cancer turned out to have abnormalities in genes not typically associated with breast cancer, including one that activates a cancer-associated protein called mTOR. The drugs were blocking the growth-stimulating activity of that protein. James’s doctor explained to her that the precise targeting of the genes in her tumor, confirmed by sequencing, were aiding her treatment.
‘Back to Normal’
“When the genome study came back, she explained how DNA is in every cell in my body,” James recalled. “There was a deficiency in the tumor, and the drugs countered that deficiency and brought me back to normal.”
More than a year later, the breast cancer hasn’t returned. However, James’s doctors found colonies of cancer cells in her liver, which they are treating with another chemotherapy regimen that combats her tumor’s inability to repair breaks in DNA. For her, the fight to find the right treatment for her disease goes on. Yet the results of her sequencing may help other women get targeted treatment for their cancers, as more doctors begin scrutinizing genes early in the disease.
“We’re not all the same,” she said. “Instead of each of us being a guinea pig, now you can tell me specifically, this is what we’re going to give Kristal, and this is what we’re going to give someone else. I think we’re making huge strides.”
The genome is the biological code that tells cells how to make their own building-block proteins. Thousands of people are diagnosed annually with illnesses clearly rooted in genes, such as Huntington’s disease, muscular dystrophy and Down syndrome.
Some genetic diseases are well understood and treatable if caught early enough. Other gene variants, like BRCA1 and BRCA2, raise the risk of breast and ovarian cancer, yet don’t guarantee that tumors will appear.
Personal Genome Project
To get a better understanding of the link between the genome and health, I joined the Personal Genome Project, run by Harvard University geneticist George Church. A genetic counselor took a history of diseases -- most of them relatively common -- that afflict people in my family.
While I waited to see what my genome can reveal about my future health, I thought about whether I might face a heightened risk of cancer, a common condition that can sometimes be staved off with diet, exercise and other health habits. Or was I destined to develop a rare condition over which I would have no control, such as Huntington’s disease?
(The sequencing results of reporter John Lauerman will be detailed later in this series.)
Dana Nieder faced a similar lack of control in her search for answers about her daughter, Maya.
Quest for Understanding
Her quest for understanding began on the day Maya was born, May 30, 2008. The infant had passed all her prenatal screening tests, which included Down syndrome and other genetic disorders, with flying colors. While Maya’s eyes were slightly wide-set and her ears low, both sometimes signs of genetic disease, doctors dismissed their concerns when she behaved like a normal newborn.
Nieder quickly discovered Maya was unable to breast-feed, apparently because of poor control of her tongue. Tests for Down syndrome came back negative. By June of 2009, when Maya was a year old, her developmental problems became more visible. The tests went on and on, yet they failed to shed a ray of light on Maya’s condition. She became more wary of doctors, who always wanted one more blood sample or one more scan, Nieder said.
“There’s the guilt of so many blood tests and so many appointments and so much for a small child,” she said.
Helping Maya
At 3 1/2 years old, Maya has just a few syllables of speech. She walks jerkily and her muscle tone remains poor. Nieder left her teaching job to stay at home with Maya and began a blog in which she describes the joys of raising her daughter and her frustration with the lack of a diagnosis.
Without a precise condition to point to, Nieder has struggled to secure services such as feeding therapy for Maya, who still has difficulty eating. Nieder is shopping for computerized communication aids that cost as much as $10,000. Insurers will be reluctant to reimburse those, too, without a diagnosis, she said.
A reader of Nieder’s blog wrote last year to tell her about the Rare Genomics Institute at Johns Hopkins University. Set up in 2010, the institute is aimed at helping parents use the Internet to raise funds for sequencing that isn’t covered by insurance. Jimmy Lin, a medical student working toward both his MD and Ph.D. degrees who leads the group, explained to Nieder that DNA sequencing could help find the root cause of Maya’s condition.
Some Concerns
“I wasn’t sure how much thought I wanted to invest in it at first,” she recalled. “I was concerned about what the sequencing might show, and who might else have access to the data.”
Maya’s genetic information would be anonymous and confidential, Lin said. One night in July, Nieder talked it over with her husband. They continued to think about whether Maya’s data would remain private and how it might be used if it fell into the wrong hands. Before the evening was out, they decided to proceed. The possibility of helping Maya even in some small way outweighed the potential dangers, which seemed vague and distant.
Maya’s evaluation took place Nov. 30 at Yale University. Geneticists recommended that she and her parents have all their protein-making genes deciphered, a process called full exome sequencing. Sequencing just Maya’s protein-making genes might answer the questions about her condition and, if that fails, the full genome could still be scanned.
First on the Block
The most difficult part of the visit to Yale, Nieder recalled, was the long recap of Maya’s health history and discussion of sequencing with William Graf, the pediatrician who took her case.
“He told us that we were doing exactly the right thing,” Nieder recalls. “He said, ‘I’d do the same thing if I were you. Everyone will be doing this soon, and you’re just the first people on the block.’”
To Maya, it was just another in a series of interminable examinations. Tired of sitting in the small room, Maya burst out of the examination room and ran around the halls of the clinic. Nieder and her husband took turns playing with Maya and speaking with Graf.
At the end of the consultation, clinic workers took blood samples from each of the Nieders to begin the sequencing. When the family returned home, Dana went online and posted an appeal for funding for the sequencing. Within just six hours, she raised more than the $2,500 needed to pay for the procedure. The Nieders say they will donate any extra money for other children who need sequencing.
While the analysis of the family’s DNA is under way at Yale, Nieder waits. She says she hopes sequencing will at least put a name to put on her daughter’s condition.
“It’s difficult to be undiagnosed. I worry that when she turns 5 and goes to school, I’m going to have to fight for services and therapies if I have nothing to write in the chart,” Nieder said.
“I’m hopeful about it, I really am.”